一、現象
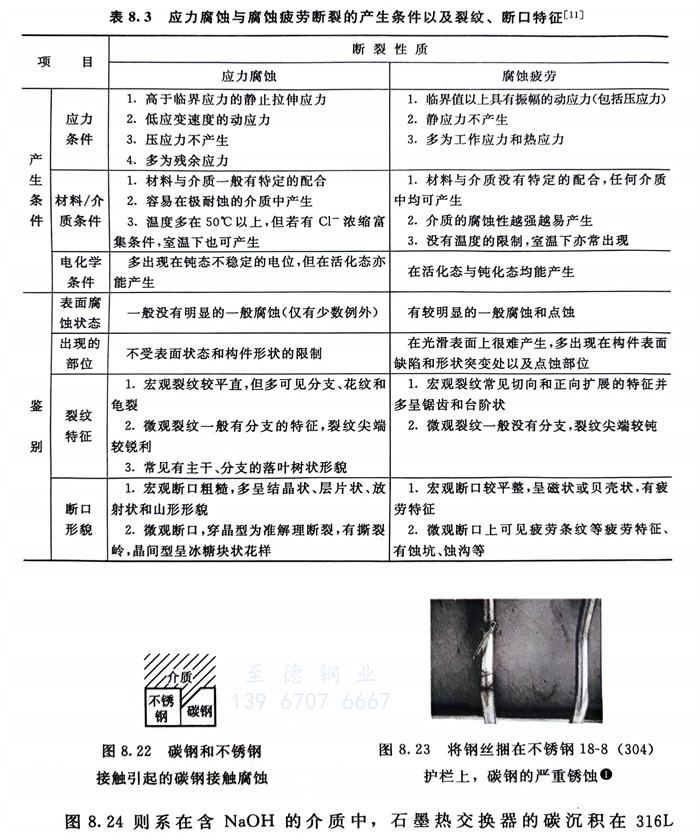
在電解質溶液中(包括潮濕大氣),碳鋼管若與不(bu)銹鋼管相接觸,碳鋼就會迅速產生腐蝕(示意圖見圖8.22),實際例子見圖8.23。后者系城市中用鋼(鐵)絲捆在18-8(304)不銹鋼護欄上所導致的鋼絲的嚴重銹蝕(鋼絲銹蝕還可誘發不銹鋼護欄的腐蝕)。
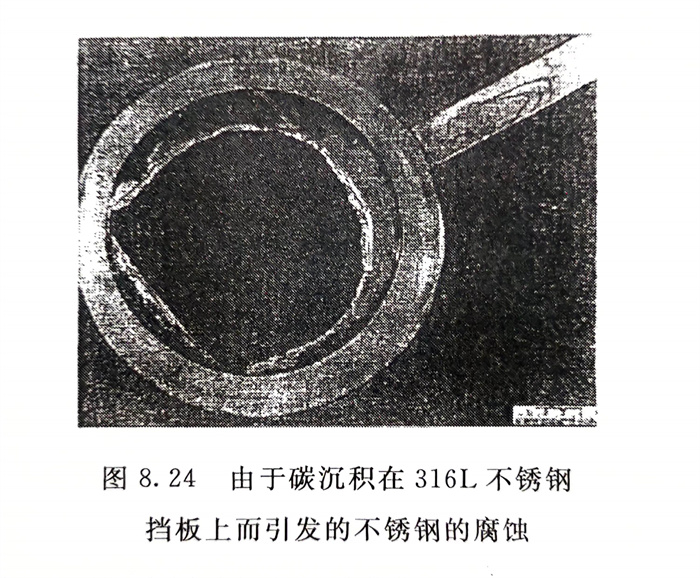
圖8.24則系在含NaOH的介質中,石墨熱交換器的碳沉積在316L(022Cr17Ni12Mo2)不銹鋼擋板上而引起的316L不銹鋼管的腐蝕。
二、原因
兩(liang)種(zhong)(zhong)金(jin)(jin)(jin)屬(shu)(shu)(shu)在腐(fu)蝕(shi)(shi)介(jie)質(zhi)中(zhong),由(you)于二者的(de)電(dian)(dian)(dian)極(ji)(ji)電(dian)(dian)(dian)位(wei)(wei)(wei)不(bu)同,電(dian)(dian)(dian)位(wei)(wei)(wei)較(jiao)(jiao)正金(jin)(jin)(jin)屬(shu)(shu)(shu)為(wei)(wei)陰(yin)極(ji)(ji),電(dian)(dian)(dian)位(wei)(wei)(wei)較(jiao)(jiao)負的(de)金(jin)(jin)(jin)屬(shu)(shu)(shu)為(wei)(wei)陽極(ji)(ji),當兩(liang)種(zhong)(zhong)金(jin)(jin)(jin)屬(shu)(shu)(shu)接(jie)觸(chu)時(shi),它們之(zhi)間就會有電(dian)(dian)(dian)流產生(sheng),出(chu)現(xian)電(dian)(dian)(dian)化(hua)學腐(fu)蝕(shi)(shi)(實際上(shang)是形(xing)成了宏(hong)觀電(dian)(dian)(dian)池(chi))。此時(shi)作(zuo)為(wei)(wei)陽極(ji)(ji)的(de)金(jin)(jin)(jin)屬(shu)(shu)(shu),其(qi)腐(fu)蝕(shi)(shi)速(su)度就會提高(加速(su)腐(fu)蝕(shi)(shi)),而(er)作(zuo)為(wei)(wei)陰(yin)極(ji)(ji)的(de)金(jin)(jin)(jin)屬(shu)(shu)(shu)其(qi)腐(fu)蝕(shi)(shi)速(su)度就會降(jiang)低(受到保護),從而(er)形(xing)成異金(jin)(jin)(jin)屬(shu)(shu)(shu)接(jie)觸(chu)(電(dian)(dian)(dian)偶)腐(fu)蝕(shi)(shi)。前述兩(liang)例(li),前者舉例(li)表明,碳(tan)鋼的(de)電(dian)(dian)(dian)極(ji)(ji)電(dian)(dian)(dian)位(wei)(wei)(wei)遠低于不(bu)銹(xiu)鋼,而(er)后者則系碳(tan)(石墨)的(de)電(dian)(dian)(dian)極(ji)(ji)電(dian)(dian)(dian)位(wei)(wei)(wei)又遠較(jiao)(jiao)316L不(bu)銹(xiu)鋼為(wei)(wei)正,從而(er)產生(sheng)了異金(jin)(jin)(jin)屬(shu)(shu)(shu)接(jie)觸(chu)腐(fu)蝕(shi)(shi)所(suo)致。
三、防(fang)止措施(shi)
在(zai)濕態腐蝕環(huan)境中(zhong),當不(bu)銹(xiu)鋼(gang)管與其他金屬(shu)相(xiang)接觸時,要(yao)考慮(lv)它們之間的電(dian)極(ji)電(dian)位(wei)是(shi)否存在(zai)顯著(zhu)的差異。不(bu)要(yao)選用與不(bu)銹(xiu)鋼(gang)相(xiang)比較電(dian)極(ji)電(dian)位(wei)相(xiang)差過(guo)大(da)的金屬(shu)。



